The Pre-Bond Convention
In creating 'pre-bonds', I sought an unambiguous use of curved arrows. I wanted to discourage students from starting curved arrows with electrophiles, like protons. I also wanted students to be able to predict the products of reactions from the curved arrows. In order to do that, I needed to make the curved arrow convention do two things. The curved arrows must show which electrons are moving and what bonds are made or broken.
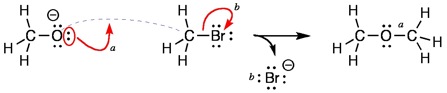
I wanted curved arrows to always be consistent. Thus, a curved arrow must start with a pair of electrons and end either between two atoms (to make a bond) or on an atom (to break a bond). In order to make predicting the product of a reaction unambiguous, I adopted the 'pre-bond' (a dashed line) to represent where a new bond will form.
In the example below, we may write the following sentences to represent the electron movements represented by the curved arrows 'a' and 'b'. For curved arrow 'a', "A new bond will be formed between the oxygen atom and carbon atom. This is the 'pre-bond' (dashed line)." The non-bonded electrons on the oxygen atoms are used to make the new bond. For curved arrow 'b', "Concurrently, the bond between the carbon and bromine
I wanted curved arrows to always be consistent. Thus, a curved arrow must start with a pair of electrons and end either between two atoms (to make a bond) or on an atom (to break a bond). In order to make predicting the product of a reaction unambiguous, I adopted the 'pre-bond' (a dashed line) to represent where a new bond will form.
In the example below, we may write the following sentences to represent the electron movements represented by the curved arrows 'a' and 'b'. For curved arrow 'a', "A new bond will be formed between the oxygen atom and carbon atom. This is the 'pre-bond' (dashed line)." The non-bonded electrons on the oxygen atoms are used to make the new bond. For curved arrow 'b', "Concurrently, the bond between the carbon and bromine
atom is broken with the electrons remaining attached to the bromine." The new bond and the non-bonded electrons that were represented by the curved arrows are noted in the products.
The issue for use of 'pre-bonds' should not be a question of whether it conforms to prior usage, but how clearly the meaning of the curved arrows is carried. In class, I found it very natural to describe bond formation with the English sentences as I indicated. I think it is easy to understand and students agree. As a result, writing reaction mechanisms with pre-bonds results in an easy to understand mechanism.
I also don't want to leave anyone thinking that I invented this use of dashed lines to show where a new bond will form. Although this use of the dashed line is not a predominant usage, it is not difficult to find either. I had searched a number of books, but I didn't make a list of sources. Diels-Alder and rearrangement reactions are likely places to find instances. For example, Williamson shows exactly that usage in Chapter 49 on Diels-Alder Reaction in Macroscale and Microscale Organic Reactions (Houghton Miflin, 4th edition).
The issue for use of 'pre-bonds' should not be a question of whether it conforms to prior usage, but how clearly the meaning of the curved arrows is carried. In class, I found it very natural to describe bond formation with the English sentences as I indicated. I think it is easy to understand and students agree. As a result, writing reaction mechanisms with pre-bonds results in an easy to understand mechanism.
I also don't want to leave anyone thinking that I invented this use of dashed lines to show where a new bond will form. Although this use of the dashed line is not a predominant usage, it is not difficult to find either. I had searched a number of books, but I didn't make a list of sources. Diels-Alder and rearrangement reactions are likely places to find instances. For example, Williamson shows exactly that usage in Chapter 49 on Diels-Alder Reaction in Macroscale and Microscale Organic Reactions (Houghton Miflin, 4th edition).