Another curved arrow example
Nov 2009
Okay, so I have my book on the market and the people I really need to adopt it are people that teach organic chemistry. However, …
Okay, so I have my book on the market and the people I really need to adopt it are people that teach organic chemistry. However, that is proving a challenge. If you read back to some of my earlier posts, you will find that one objective I had in writing my book was to eliminate ambiguity and confusion. Below is something I found on the internet. It was about SN1 v E1 reactions. I won’t post the author.

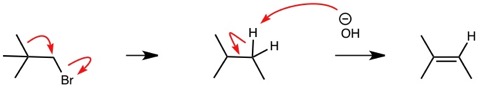
Let’s look at this again, “I just lectured on ….” So, this is someone that teaches organic chemistry. Let’s look at the curved arrows used by this person. Okay with the first set, that means rearrangement. Let’s look at the middle set. That looks like a second rearrangement, doesn’t it?
This was my point. If I was a student in this class, would I pay attention to the curved arrows or would I just ignore them? There is not anything particularly wrong with the curved arrows in this instance except they are bordering on ambiguity. I only say it is bordering on ambiguity as the pair of electrons from the (-)OH implies a bond change for the C-H. The middle set of curved arrows should have pointed to between the atoms to indicate a change in bond order.
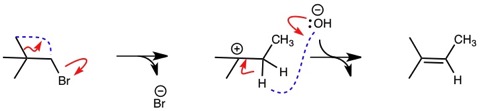
Let’s review the curved arrows with how they might have been written. I have tried to be more careful so that a curved arrow starts with a pair of electrons and ends between the atoms to change the bond order to a double bond.
You may also notice that the original curved arrow in the middle set appears as though it is being used to break a bond, like the example below.

For this reason, I had adopted a more unambiguous way of using curved arrows, see below with the same example.
… I just lectured on beta branching this morning, and neopentyl electrophiles react some 100,000 times slower than ethyl bromide in SN2 reactions. So if you let it go long enough, yeah, you'd probably get some SN2... eventually. So I predict one of the rare cases where rearrangement and LG expulsion occur simultaneously. This gives the stable tertiary carbocation. We're in good base, so E1 elimination should occur after carbocation formation. …
(My sketch of the arrow-pushing is below.)

Let’s look at this again, “I just lectured on ….” So, this is someone that teaches organic chemistry. Let’s look at the curved arrows used by this person. Okay with the first set, that means rearrangement. Let’s look at the middle set. That looks like a second rearrangement, doesn’t it?
This was my point. If I was a student in this class, would I pay attention to the curved arrows or would I just ignore them? There is not anything particularly wrong with the curved arrows in this instance except they are bordering on ambiguity. I only say it is bordering on ambiguity as the pair of electrons from the (-)OH implies a bond change for the C-H. The middle set of curved arrows should have pointed to between the atoms to indicate a change in bond order.
Let’s review the curved arrows with how they might have been written. I have tried to be more careful so that a curved arrow starts with a pair of electrons and ends between the atoms to change the bond order to a double bond.

You may also notice that the original curved arrow in the middle set appears as though it is being used to break a bond, like the example below.
For this reason, I had adopted a more unambiguous way of using curved arrows, see below with the same example.
